|
3/18/2024 0 Comments Bohr model of the hydrogen atom 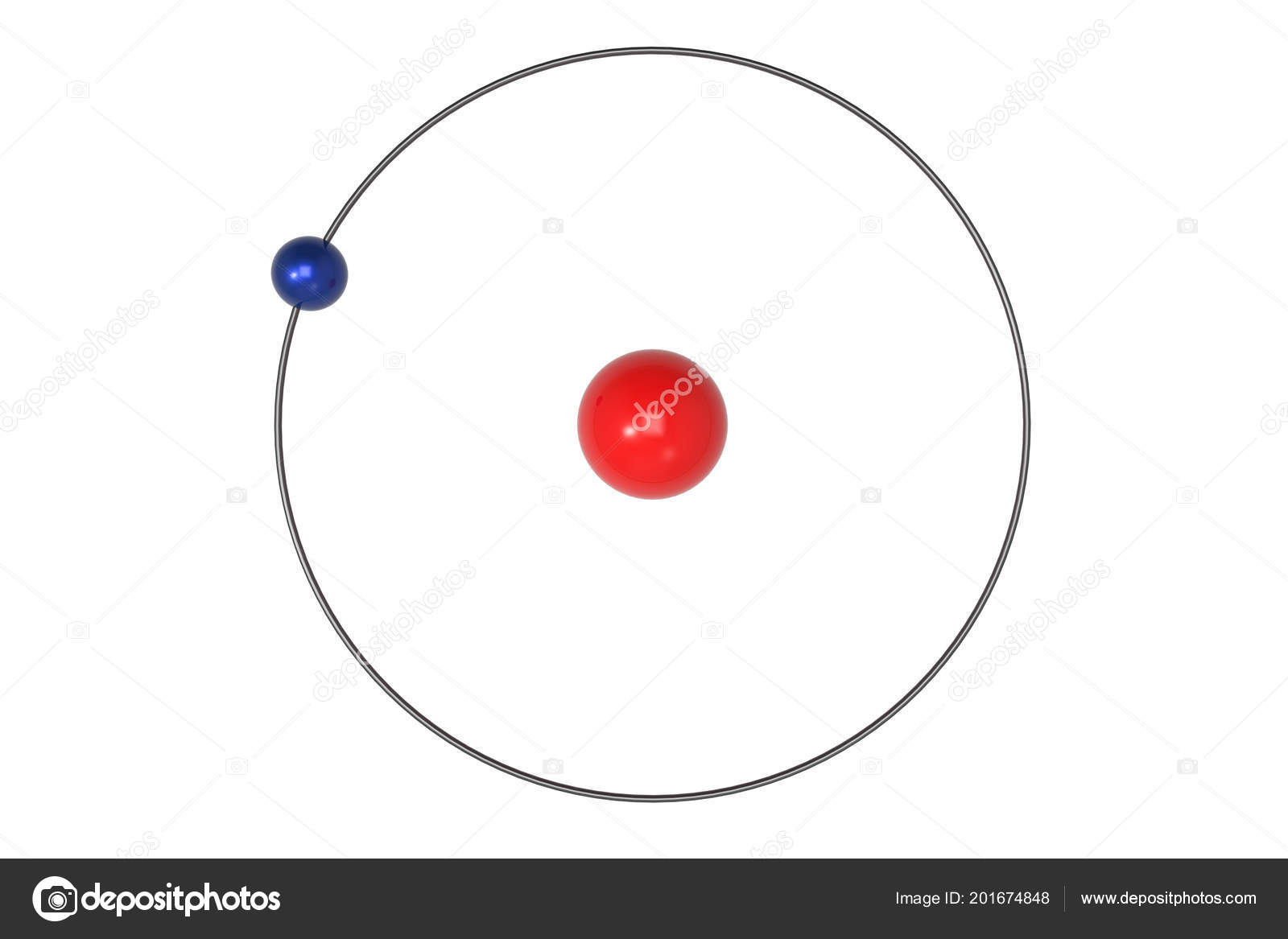
Find the radius r 1 of a valence orbital for a He+ ion. 
It does introduce several important features of all models used to describe the distribution of electrons in an atom. 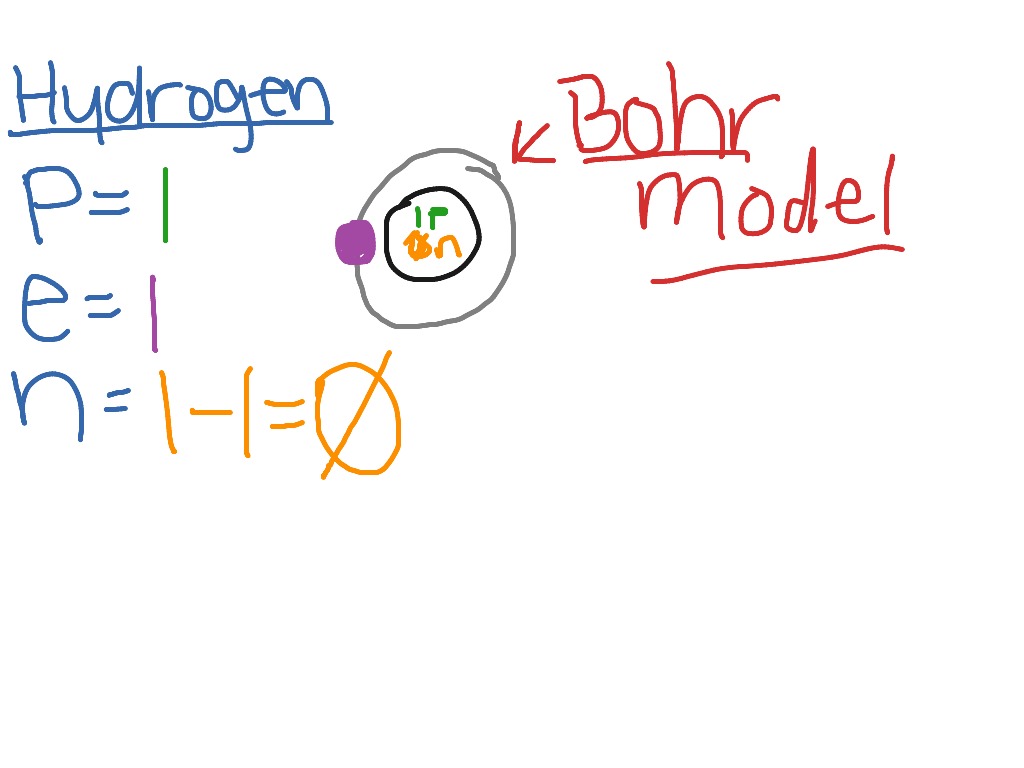
When electrons return to a lower energy level, they emit energy in the form of light. Orbits further from the nucleus exist at higher energy levels. B)In the Bohr model of hydrogen, the radius of the n th orbit is defined as. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it does not account for electronelectron interactions in atoms with more than one electron. The Bohr model postulates that electrons orbit the nucleus at fixed energy levels. A) Find the frequency of light f radiated by an electron moving from orbit n 12 to n 21 inside of a He+ ion. With his model, Bohr explained how electrons could jump from one orbit to another only by emitting or absorbing energy in. As Bohr had noticed, the radius of the n 1 orbit is approximately the same size as an atom. The quantity in parentheses is a unit of energy known as a Rydberg, which \(\approx 13.6eV\).īohr reasoned that the hydrogen atom doesn't radiate away all of its energy, because the lowest energy level (which we would now call the ground state) corresponds to one wavelength fitting in the orbit, so \(n=1\) is the lowest it can go. In the case of hydrogen ( Z 1) E 013.6eV. In Bohr’s model, radius an of the orbit n is given by the formula an h2n2 0 / 2, where 0 is the electric constant. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
